 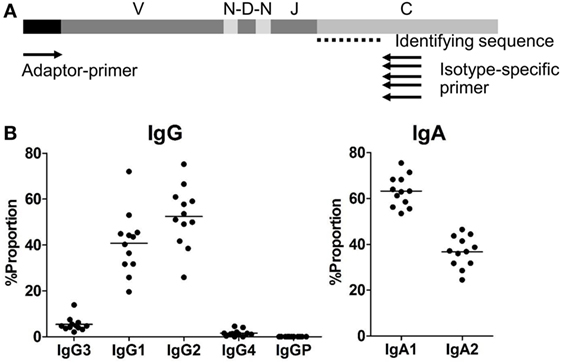
These transgenic-knockout approaches revealed that diversification and selection in integrated human Ig loci are controlled by the animal’s immune system and that they undergo natural processes of V(D)J rearrangement, somatic hypermutation (SHM) and class-switching. The first generation of humanized immunoglobulin (Ig) mice was developed in the 1990s by random integration of DNA segments containing partial human Ig heavy and light chain loci into the chromosomes of endogenous Ig-knockout mice 2, 3. Transgenic animals designed to express the human Ab repertoire are widely recognized for their ability to generate fully human mAbs 1, 2. Genetic engineering has been used to develop various methods to overcome the immunogenicity of rodent monoclonal antibodies (mAbs) in humans, which was a critical issue in early clinical trials. In the past two decades, therapeutic antibodies (Abs) have emerged as a highly effective and fast-growing pharmaceutical option. Our results thus suggest that TC-mAb mice offer a valuable platform for obtaining fully human therapeutic antibodies, and a useful model for elucidating the regulation of human Ig repertoire formation. Despite slightly altered B cell development and a delayed immune response, TC-mAb mice have more subsets of antigen-specific plasmablast and plasma cells than wild-type mice, leading to efficient hybridoma production. Comprehensive, high-throughput DNA sequencing shows that the human Ig repertoire, including variable gene usage, is well recapitulated in TC-mAb mice. Here, we establish human antibody-producing Tc mice (TC-mAb mice) that stably maintain a mouse-derived, engineered chromosome containing the entire human Ig heavy and kappa chain loci in a mouse Ig-knockout background. In this mini review, I will briefly discuss (i) our knowledge regarding the prevalence of anti-AAV antibodies, (ii) the challenges to measure those antibodies by methods that are most predictive of their influence on therapeutic efficacy of AAV gene transfer, and (iii) approaches to overcome the formidable hurdle that anti-AAV antibodies pose to the successful clinical use of AAV gene therapy.Īav adeno-associated virus antibodies antibody assays gene therapy prevalence of antibodies removal of antibodies removal of inhibitors.Trans-chromosomic (Tc) mice carrying mini-chromosomes with megabase-sized human immunoglobulin (Ig) loci have contributed to the development of fully human therapeutic monoclonal antibodies, but mitotic instability of human mini-chromosomes in mice may limit the efficiency of hybridoma production. But the successful use of AAV vectors to treat an increasing number of diseases also makes establishing protocols to determine therapeutically relevant titers of pre-existing anti-AAV antibodies and approaches to deplete those antibodies more urgent than ever. The recent tragic death of three children in a clinical trial to treat X-Linked Myotubular Myopathy by delivering myotubularin with an AAV8 vector notwithstanding, AAV remains a highly promising therapeutic gene delivery platform. Adeno-associated virus (AAV) vector-based gene therapy is currently the only in vivo gene therapy approved in the US and Europe. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
